Gamma-PET
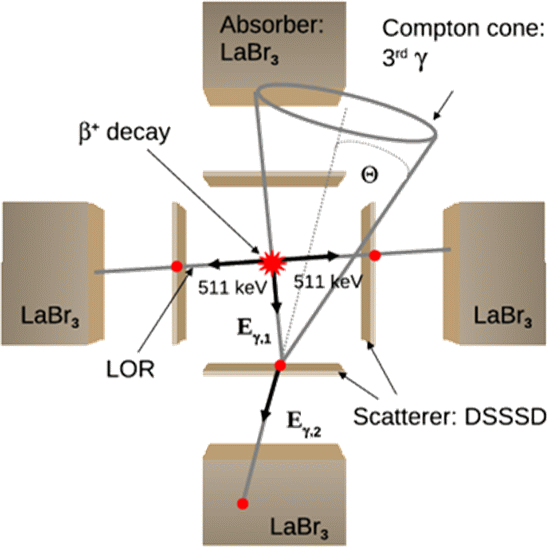
So far a whole class of potential PET isotopes, where in addition to the two back-to-back emitted 511 keV annihilation photons also a third, high-energy γ ray is emitted from an excited state in the daughter nucleus, has been excluded from medical application. The resulting extra dose delivered to the patient, as well as the expected increase of background from Compton scattering or even pair production, prevented the use of isotopes such as 44mSc, 86Y, 94Tc, 94mTc, 152Tb, or 34mCl. However, provided the availability of customized gamma cameras, this alleged disadvantage could be turned into a promising benefit, offering a higher sensitivity for the reconstruction of the radioactivity distribution in PET examinations. In addition to envisioned applications in nuclear medicine imaging, combined detection of PET and single photons could open new avenues in range verification of particle therapy, by integrating the imaging modalities of PET, prompt gamma and triple β+-γ coincidences (the latter drawing on the irradiation-induced production of isotopes such as 10C and 14O, which emit a third gamma in addition to the β+-emission).
Presently, several approaches are pursued towards the realization of a medical imaging system based on β+-γ coincidences. All of them draw on the imaging properties of a Compton camera, where the registration of the Compton scatter and absorption kinematics of an incident photon in a suitable detection system is exploited to reconstruct the source position, within one event restricted to the surface of a cone. Determining the intersection of this Compton cone with the line-of response (LOR) as defined by the positron annihilation allows for a sensitive reconstruction of the decay position of the PET isotope. The above schematic figure illustrates the γ-PET technique, here based on the use of 4 Compton-camera arms. Another option, with improved reconstruction efficiency, would be to combine a conventional PET scanner with an inner ring of scatter detectors, as presently investigated by our Japanese collaborators at QST-NIRS in Chiba. Additional possibilities are being explored in the framework of two third-party funded projects supported by the EU and the Humboldt Foundation, again relying on the codes Geant4 and an experimental version of MegaLib, recently extended by its main developer to support all modalities of Compton, PET and β+-γ imaging.
Contact:
Prof. Katia Parodi
PD Dr. Peter Thirolf
References:
- C. Lang, D. Habs, P.G. Thirolf, A. Zoglauer, Radiotherapy and Oncology 102, s1 (2012), S29.
- C. Lang, D. Habs, K. Parodi, P.G. Thirolf, Jour. of Instrumentation 9 (2014) P01008.
- P.G. Thirolf, C. Lang and K. Parodi, Perspectives for highly-sensitive PET-based medical imaging using β+γ coincidences, Acta Physica Polonica A 127, 1441 (2015).
Currently funded projects:
- 2018-2021, International Research Initiative, funded by QST-NIRS, PI at QST-NIRS: T. Yamaya, PI at LMU: K. Parodi
- 2017-2019, Hybrid Imaging of PET and PrOmpt gamma for preCision RAnge- and biological- guidance in proton ThErapy (HIPPOCRATE), funded by EU via Marie Sklodowska-Curie individual fellowship, PI: V. Anagnostatou, Coordinator: K. Parodi
- 2018-2020, Humboldt Fellowship, funded by Humboldt Foundation, PI: M. Safari

