Ionoacoustics
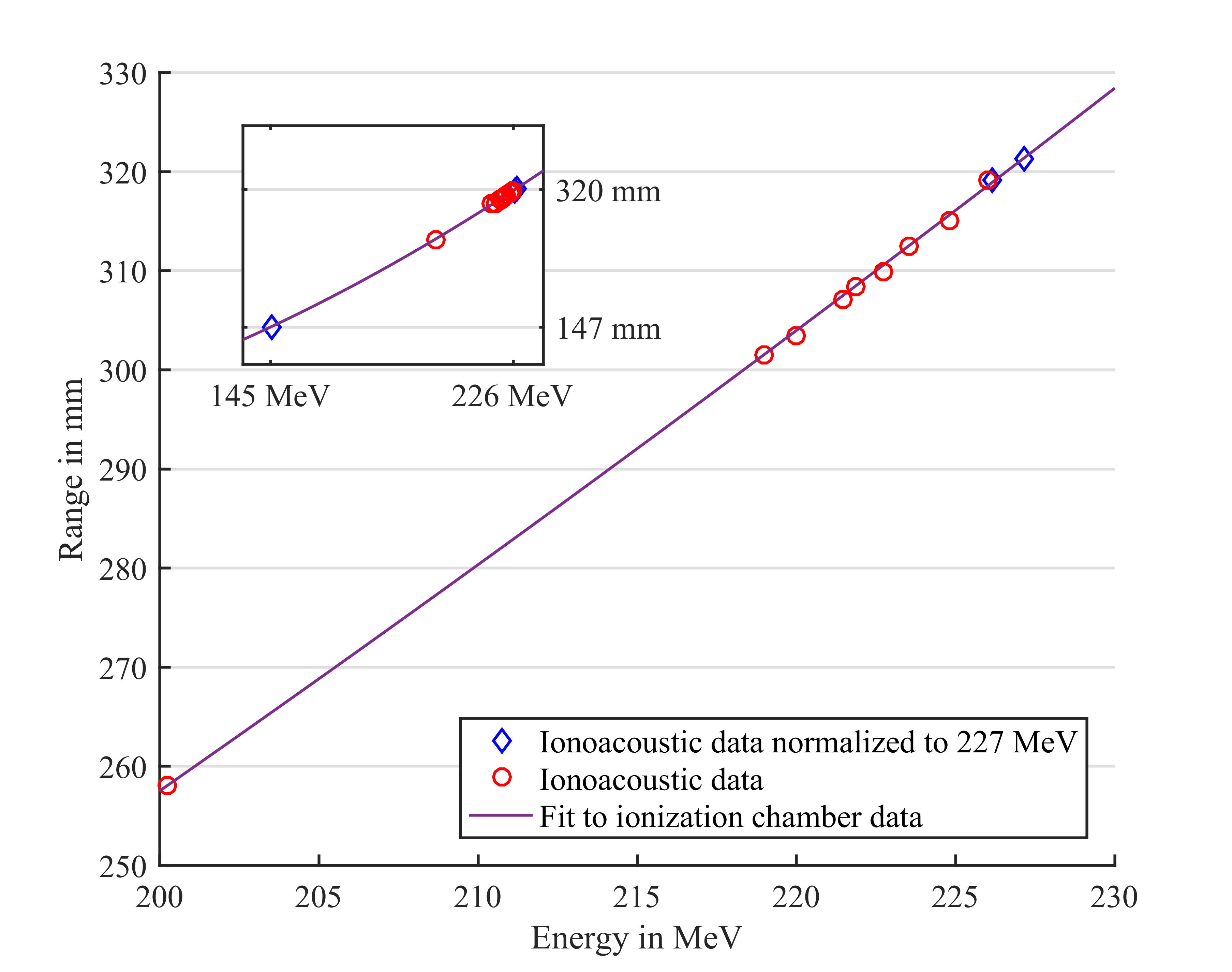
When penetrating a medium, ions mainly lose energy in electronic collisions, resulting in localized heating and a thermal expansion, which generates thermoacoustic emissions detectable with acoustic transducers. Being the thermoacoustic emission naturally enhanced at the maximum of ion energy deposition (so-called Bragg-peak), time-of-flight (TOF) measurements in combination with knowledge of the speed of sound in the traversed medium can enable recovery of the ion range. Especially in combination with morphological ultrasonography, this so-called “ionoacoustics” could offer a compact and cost-effective modality for real-time in-vivo verification of the beam range, co-registered to tissue anatomy, at least for suitable indications of feasible sonic access [1]. However, strength and shape of the ionoacoustic signal crucially depend on the spatial distribution and temporal structure of the heating process, i.e., dose deposition. For a deeper understanding, we extensively characterized ionoacoustic emissions produced by pulsed proton beams in water, at pre-clinical energies delivered by the Tandem accelerator of the Maier Leibnitz Laboratory [2] as well as at clinical energies delivered by the proton synchrocyclotron at the collaborating Center Antoine Lacassagne in France, Nice [3]. In the considered scenario of axial detection of ionoacoustic emissions distal to the Bragg-peak, all performed TOF measurements indicated the potential of (sub)millimetre accuracy and precision for range monitoring (figure 1,2), provided that sufficient signal was collected, e.g., by averaging over more proton pulses for total doses up to 10 Gy. While the considered very low beam energies of 20 MeV generated high-frequency acoustic waves detectable with typical ultrasound transducers operating in the MHz range, clinical beam energies up to 227 MeV delivered by the intrinsically pulsed synchrocyclotron demanded the usage of broadband, low-frequency (kHz) hydrophones. Simulations studies currently extending the experimental findings in water to more realistic heterogeneous tissue indeed support our ongoing efforts to translate ionoacoustics into a pre-clinical and clinical proton range monitoring technique. To this end, our main lines of research aim at:

- Developing an optoacoustic setup to emulate the signal produced by ion beam interactions, to provide a test bench for optimization of detectors and imaging approaches,
- Harnessing detector technologies for application to a novel small animal proton irradiation, ideally combining ionoacoustic detection with ultrasonography,
- Improving the signal-to-noise ratio by enhancing detector performances and signal acquisition, to pave the way toward future clinical application at intrinsically or artificially pulsed proton beams.
Contact:
Dr. Julie Lascaud
Prof. Dr. Katia Parodi
References:
[1] K. Parodi and W. Assmann 2015 Mod. Phys. Lett. A 30 1540025
[2] W. Assmann et al 2015 Med. Phys. 42 567–74
[3] S. Lehrack et al 2017 Phys. Med. Biol. 62 L20
[4] S. Lehrack et al. 2020 Nucl. Instr. Meth. A 950 162935
Currently funded projects:
- 2020-2022, Ionoacoustics@GSI, funded by GSI via F&E collaboration, PI: W. Assmann, K. Parodi (LMU), Chr. Trautmann (GSI)

